
While it is more appropriate to refer to equivalent weight as equivalent mass, this term is rarely used. Because 1 mole of acid or base is equal to 1 equivalent of acid or base, equivalent weight is expressed in grams per equivalent (g/equivalent or g/eq) (Equation 2). Another way of expressing this is that the equivalent weight is defined as the mass (in grams) of a base that reacts with exactly 1 mole of hydrogen ions (H +). For bases, the equivalent weight is defined as the mass (in grams) of a base that provides exactly 1 mole of hydroxide ions (OH −) to a reaction. Another way of expressing this is that the equivalent weight is defined as the mass (in grams) of an acid that reacts with exactly 1 mole of hydroxide ions (OH −). For acids, the equivalent weight (EW) is defined as the mass (in grams) of an acid that provides exactly 1 mole of hydrogen ions (H +) (to be precise, hydronium ions, H 3O +) to a reaction. Similar to molarity, where the molecular weight (MW) is used for calculating the molar concentration, for normal solution concentrations, it is desirable to use the equivalent weight (EW). Thus, in order to have a one-to-one relationship between acids and bases, many chemists prefer to express the concentration of acids and bases in normality. Normality is used in place of molarity because often 1 mole of acid does not neutralize 1 mole of base. Normality is defined as the number of equivalent weights (or simply equivalents, eq) of solute dissolved per liter of solution (equivalents/L = N) (Equation 1). It is not flammable.In some cases, particularly in situations involving acid-base chemistry, the solution concentration is expressed in normality (N or C N). Its ingestion can cause somnolence, delirium and psychosis. Health effects / safety hazards: Potassium bromide is an irritant to eyes. Potassium bromide is added in some formulation to reveal photographies. It is commonly used in the technique of Infrared spectroscopy due to the crystal formed is transparent and does not have optical absorption. 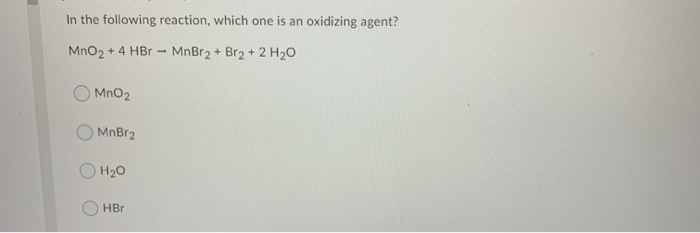
Today, it only remains as medicine to treat epilepsy of veterinary use. Uses: Potassium bromide was used as medicine to combat the convulsion in century XIX and first part of XX. KBr(aq) + AgNO 3(aq) → AgBr(s) + KNO 3(aq) The formation of bromine anion can be used to produce some salts as silver bromide, which precipitates in water and can be recovered through filtration: Due to great difference the ionic bond is stronger but at the same time, the molecule can easily dissociate in water to form the ions. It is hygroscopic.Ĭhemical properties: Potassium bromide is formed by two ions with high difference of electronegativity: potassium cation K + and bromine anion Br. Potassium bromide is highly soluble in water and slightly soluble in diethyl ether. The melting point of the salt is 734 ☌, while the boiling point is 1435 ☌. Physical properties: Potassium bromide is an odorless, white crystalline solid, with a sweet taste and with a density of 2.74 g mL -1. Preparation: Potassium bromide is prepared by the reaction of potassium carbonate with iron (III) bromide:Ĥ K 2CO 3 + Fe 3Br 8 → 8 KBr + Fe 3O 4 + 4 CO 2 Occurrence: Potassium bromide is not found in nature. Its chemical structure can be written as below, in the common representations used for organic molecules. The crystalline structure is an octahedral formed by one bromine anion surrounded by six potassium cation and vice versa. The structure of the salt is formed by one cation K + and one anion Br. Formula and structure: Potassium bromide chemical formula is KBr and the molar mass is 119.00 g mol -1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
